Feb. 28, 2026
Full Traceability with ISO 9001 & ISO 13485 Certification in Sheet Metal Fabrication
In precision sheet metal fabrication, full traceability is not optional — it is a core requirement for industries where safety, functionality, and regulatory compliance are directly tied to how each part is produced, inspected, and documented.
At Rollyu Precision, full traceability is fully integrated into every stage of our manufacturing process — from incoming raw materials to finished product delivery — in full compliance with both ISO 9001:2015 and ISO 13485:2016 certifications.
ISO 9001 and Traceability in Sheet Metal Fabrication
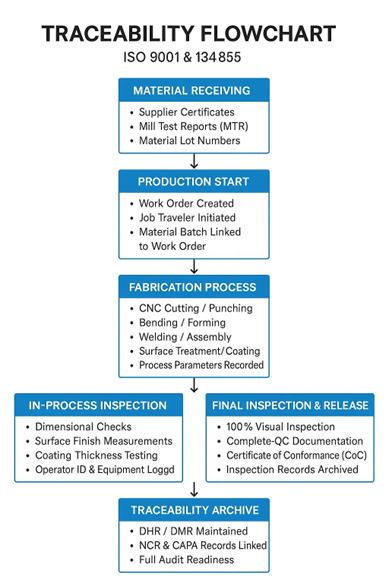
ISO 9001:2015, the globally recognized standard for quality management systems, addresses traceability requirements under Clause 8.5.2 – Identification and Traceability. While ISO 9001 allows for flexibility depending on customer and industry needs, full traceability systems in sheet metal fabrication typically include:
Product Identificatio Full Traceability with ISO 9001 & ISO 13485 Certification in Sheet Metal Fabrication
In precision sheet metal fabrication, full traceability is not optional — it is a core requirement for industries where safety, functionality, and regulatory compliance are directly tied to how each part is produced, inspected, and documented.
At Rollyu Precision, full traceability is fully integrated into every stage of our manufacturing process — from incoming raw materials to finished product delivery — in full compliance with both ISO 9001:2015 and ISO 13485:2016 certifications.
n: Each part, batch, or work order carries unique identifiers (lot codes, serial numbers, job travelers).
Material Traceability: Mill certifications (MTRs), supplier certificates, and batch records link raw material batches to each fabricated part.
Process Documentation: Every step in the production sequence — cutting, bending, welding, finishing, coating — is recorded with operator ID, machine ID, and timestamp.
Inspection Records: Dimensional checks, surface finish inspections, coating measurements, and final quality control results are documented for each batch.
Non-Conformance Tracking: Any deviations are logged, investigated, and resolved under documented corrective action procedures (NCR, CAPA).
Audit-Ready Documentation: Complete traceability packages are maintained for customer audits, internal reviews, and regulatory inspections.
While ISO 9001 applies across many industries, this level of traceability provides the foundation for highly regulated sectors that demand even stricter process control.
ISO 13485 and Traceability for Medical Device Components
For medical device applications, ISO 13485:2016 establishes far more rigorous traceability standards, as required by FDA regulations, EU MDR, and other global health authorities. In precision sheet metal fabrication, ISO 13485 compliance ensures that fabricated components meet strict regulatory expectations for:
Device-Level Traceability: Each component can be traced not only to its material and manufacturing process but also to its final device assembly.
Component Lot Control: All material batches, process records, and inspection data are tied to specific device lots or serial numbers.
Process Validation: Critical manufacturing processes such as welding, surface finishing, and coating are validated, controlled, and monitored.
Device History Records (DHR): Complete documentation files are maintained for each device lot, including materials, processes, inspections, and certifications.
Risk Management: Full documentation ensures that any product issues can be rapidly investigated and contained, with full transparency across the entire supply chain.
Corrective and Preventive Action (CAPA): ISO 13485 requires that all non-conformances are systematically analyzed, root causes identified, and permanent corrective actions implemented.
The Value of Implementing Full Traceability
For companies operating under ISO 9001 and ISO 13485 certifications, full traceability delivers clear, measurable benefits:
✅ Regulatory Compliance — Satisfies FDA, EU MDR, and global customer requirements.
✅ Rapid Problem Resolution — Immediate lot-level tracking for root cause analysis and containment.
✅ Supply Chain Confidence — Enables end-to-end documentation from raw material supplier to finished product delivery.
✅ Audit Readiness — Traceability packages are always prepared for both customer and regulatory audits.
✅ Customer Satisfaction — Builds trust by demonstrating full manufacturing control.
✅ Business Growth — Opens access to regulated markets including medical devices, life sciences, semiconductors, photonics, and aerospace.
Full Traceability in Action at Rollyu Precision
At Rollyu Precision, full traceability is not limited to documentation alone — it is embedded into every operational layer of our precision sheet metal fabrication process:
Raw material batch control with MTR linkage
Work order travelers linked to operator, equipment, and process history
In-process inspection data capture at every critical stage
Process validation files for coating, welding, surface finishing, and critical tolerances
Surface finish inspection reports, Ra roughness measurements, coating thickness logs
Full inspection record retention and customer access to certification packages
Complete non-conformance investigation and corrective action tracking
DHR and DMR archives for regulated device production
ISO 9001:2015 and ISO 13485:2016 fully certified system oversight
Conclusion
Full traceability is not simply a document — it is a system-wide discipline.
Through ISO 9001 and ISO 13485 certified processes, Rollyu Precision provides customers with the confidence that every fabricated part is built, inspected, and documented to meet the most demanding quality and regulatory requirements — across medical devices, photonics, semiconductors, aerospace, and automation industries.
Need full material-to-device traceability for your next precision sheet metal project?
Partner with Rollyu Precision — ISO 9001:2015 & ISO 13485:2016 certified — and ensure every part is documented, controlled, and audit-ready.
Previous: FAQs for Sheet Metal Projects
Next: Surface finish standards